DESULPHURIZATION OF PETROLEUM PRODUCTS AND SEWAGE DECONTAMINATION USING NEW ADVANTAGES OF HETEROGENEOUS CATALYST
A.G. Akhmadullina, R.M. Akhmadullin
KAZAN NATIONAL RESEARCH TECHNOLOGICAL UNIVERSITY
Abstract
Comparative analysis of homogeneous-catalytic and heterogeneous-catalytic processes at sweetening (demercaptanization) light hydrocarbons, kerosene, gas condensates and light oils, as well as disposal of sulfur-alkaline wastes. The advantages of the proposed heterogene-ous polymer-based KSM catalyst. Ways of reducing total sulfur content in light oils and gas condensates.
1. Light hydrocarbons demercaptanization (sweetening)
Light hydrocarbons purification is based on mercaptans extraction using alkaline solution with the following regeneration by oxidation of alkali mercaptides to disulfides in the presence of a catalyst:
NaOH + RSH ? RSNa + H2O (1) – extraction
2 RSNa + 0.5O2 + H2O = RSSR? + 2 NaOH (2) – regeneration
Disulfides formed are not soluble in an alkaline solution. They are removed from the regenerated alkali by gasoline extraction.
When using homogeneous catalysts dissolved in alkaline the formation of disulfide extends beyond the rege-nerator – in pipelines or in the extractor. Disulfides formed are moved in an extractor from circulating alkaline solu-tion to cleaning light hydrocarbons and therefore increasing the total sulfur content in the purified product.
Butane-butylene fraction (BBF) of the Ufa Refinery is treated in the presence of an alkaline solution of cobalt sulfoftalotsianine up to a residual content of mercaptan sulfur 5 ? 12 ppmw, but due to the presence of about 0.0195% weight disulfides in the regenerated alkaline solution entering the extractor total sulfur content in BBF is 63 ?89 ppmw.
The same problem was observed at the Ryazan Refinery while sweetening BBF performed in the presence of an IVKAZ alkaline catalyst solution (ACS). In BBF, cleaned from mercaptan sulfur up to 0.0002% by weight, was contained up to 0.002% sulfur disulfide. It was found that in the regenerated ACS solution immediately af-ter cleaning form disulfides the content of mercaptides was higher by 30 ? 50% than at the entrance of ACS in the extractor. This confirms the occurrence of mercaptides oxidation in the alkaline solution of ACS outside the rege-nerator (in the pipeline before the extractor).
Similar results using a process with a water-soluble homogeneous catalyst have been obtained in Iranian Re-fineries. In sweetened propane and butane fractions out of mercaptans up to ? 5ppmw total sulfur content found by the international standard EN 589-2008 was 50ppmw. To reduce total sulfur content up to ? 35ppmw these fractions were subjected to advanced adsorption treatment using zeolites.
Due to the tightening of regulations on the sulfur content of automotive fuels to ? 50ppmw Euro-4 and to ? 10ppmw Euro-5, as well as high-octane additives to them that synthesized from liquefied gases, regeneration of al-kaline solution is appropriate to maintain using heterogeneous catalyst, permanently fixed in the regenerator.
For this purpose we have developed polymeric KSM catalyst that produced in the form of packed elements with an exposed surface. The active components of KSM catalyst are solidly supported on polymer, which excludes them from dissolving in the circulating alkaline solution and the formation of disulfides outside the regenerator.
Using this catalyst, we have developed “Demer-LUVS” process for liquefied gas purification (Fig.1), which is provided on Ma?eikiai, Moscow, Ufa, Lisichansk, Omsk Refineries for sweetening BBF, the Novo-Yaroslavl and Nizhnekamsk Refineries – to clean propane-butane-butylene fraction (PBBF) and JSC “LUKOIL-NizhegorodNOS” – to clear butane fraction (BF).
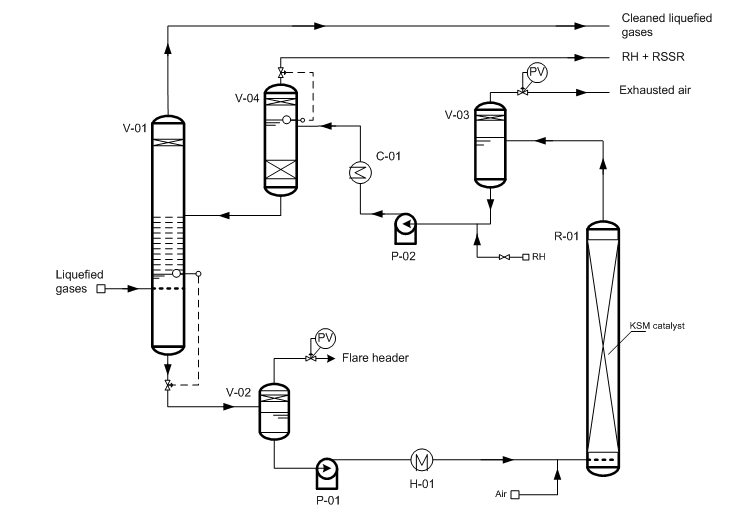
Fig.1 “Demer-LUVS” – light hydrocarbons sweetening process.
(V-01) – extractor; (V-02) – degasser; (V-03) – degasser; (V-04) – settler; (R-01) – regenerator; (P-01/02) – pump; (H-01) – heater; (C-01) – cooler.
Experience at the refinery commercial operation has shown the following significant advantages over its ho-mogeneous-catalytic processes:
• Reduction of total sulfur in the purified product less than 10ppmw by eliminating contamination of disulfides.
• Reduce operating costs by increasing the lifetime of the alkaline solution from 3 months to 1 year and lifetime of the catalyst from 3 months to 10 years.
• Sweetening units’ environmental improvements by reducing the volume of sulfur-alkaline waste, re-ducing the concentration of free alkali and getting an exception of heavy metals in waste water.
Thus, in BBF, sweetened from mercaptans by Demer-LUVS at JSC “TAIF-NK”, mercaptan sulfur contents is 1 ? 5 ppmw, and total sulfur content – does not exceed 10 ppmw. Similar results were obtained in JSC “LUKOIL-NizhegorodNOS” the content of mercaptan sulfur in BF – less than 5 ppmw, and total sulfur found by the ANTEK de-vice less than 10 ppmw.
2. Kerosene (jet fuel) fractions demercaptanization
With the putting into operation of hydrocracking refineries there were significant resources of desulfurized kerosene fractions. This makes it possible to increase the production of jet fuel due to the involvement of vir-gin kerosene fraction by manufacturing composite jet fuel TS-1 or Jet A-1. To involve the entire volume of virgin ke-rosene in the composite jet fuel it is needed to sweeten it from mercaptan sulfur content, which is 4 ? 10 times higher than normal, acceptable to jet fuel – 0.003 wt%.
In this context, becomes actual the process of selective oxidation of corrosive mercaptans into inert disulfides in alkaline medium at 40 ? 60°C. Estimated capital cost for hydrotreatment of kerosene compared with alkaline swee-tening is higher 10 ? 20 times, and operating costs – 20 ? 50 times.
The main obstacle to widespread adoption of the known processes of alkaline kerosene sweetening using based on coal catalysts – is their multistage and ecology. The fragility of the adsorption interaction of porous carbon with an alkaline solution of ACS leads to constant erosion of the long carrier. This calls for constant feeding of coal and cleaning the fuel from the ACS, which leads to the formation of the multistage waste at all stages of jet fuel purifica-tion (Fig. 2):
– Alkaline cleaning of kerosene from acidic impurities – 18 L. Sulfur alkaline waste (SAW)/tn.
– Kerosene water flushing from blown ACS – water consumption 18 L./tn.
– Kerosene salt drying from moisture and traces of ACS – salt consumption 0.26 kg/tn.
– Bleaching clay purification from traces of ACS and resins ? 95 tn./year to dump.
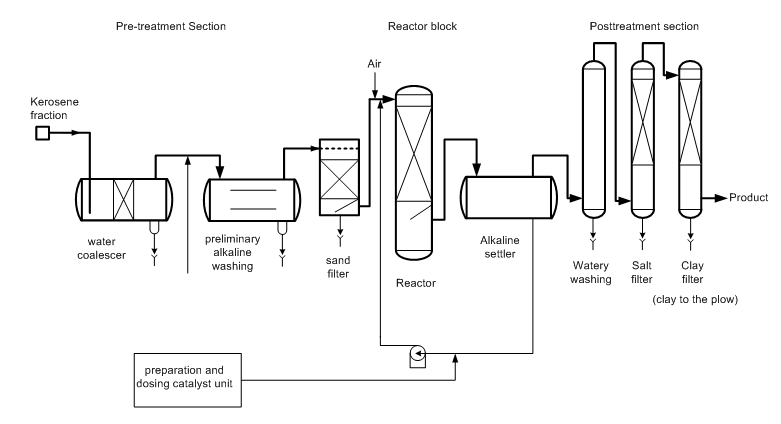
Fig. 2 Typical kerosene sweetening process using the coal based catalyst.
Instead, these processes, we propose two-stage regenerative low-waste “Demer-KSP” kerosene treatment process using polymer KSM catalyst in conjunction with the promoter that can quantitatively settle from kerosene in 60 ? 90 min. In this case, together with sweetening, in the same reactor promoter is extracting acidic impurities from kerosene, as well as part of the reaction and dissolved water, which are then picked out while promoter regenera-tion (Fig. 3).
Significant advantages of the “Demer-KSP” process using KSM catalyst and promoter compared with listed above processes are:
• Reduction of capital and operating costs for jet fuel cleaning due to the exclusion the following units:
– Preliminary alkaline treatment of kerosene form acidic impurities.
– Sweetened jet fuel water washing from traces of ACS.
– Kerosene moisture salt drying.
– Kerosene bleaching clay adsorptional purification from traces of copper salts.
• Improving ecology of the Refinery by eliminating of alkali naphthenate toxic wastes, cobalt salts and clay slurry.
Experimental batch of virgin kerosene, sweetened by pilot plant of the Moscow Refinery using “Demer-KSP” method, has successfully passed the qualification tests in VNIINP (All-Russian Research Institute for Oil Refining). With three major Refineries in Russia concluded license agreement for the implementation of this process.
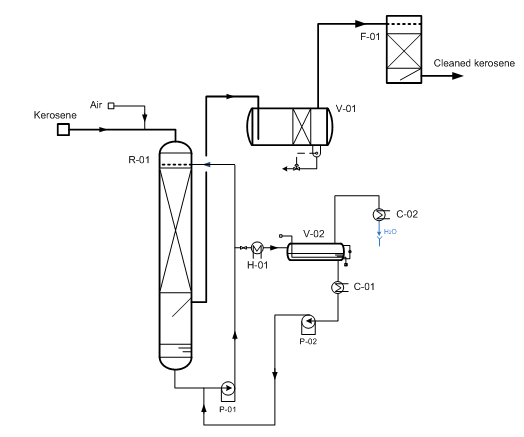
Fig. 3 Sweetening kerosene fractions using “Demer-KSP” method.
(R-01) – reactor; (V-01) – settler; (V-02) – evaporator; (P-01/02) – pumps; (H-01) – heater; (C-01/02) – cooler (F-01) – sand filter.
3. Demercaptanization of gas condensates and light oils
During hydrotreatment of Astrakhan high mercaptan gas condensate was established that the presence of mercaptans in the feed leads to increase the frequency of regeneration. The reason is the intensive formation of coke deposits by thermal decomposition of mercaptans with polymerization products of their decomposition on the catalyst. The share of these products up to 60% by weight of coke deposits.
These data indicate the feasibility of pre-sweetening high mercaptan raw before hydrotreatment. This scheme is attractive in terms of reducing the corrosivity of the material in case of mercaptans removal that caus-ing intense corrosion of equipment in the heating zones due to the mercaptans low threshold of thermal stability (140 ? 170°C).
The share of mercaptan sulfur has a number of fields from 14 to 67% of the total sulfur content (Table 1).
Table 1. The content of total and mercaptan sulfur in the oil and gas condensate depending on the location.
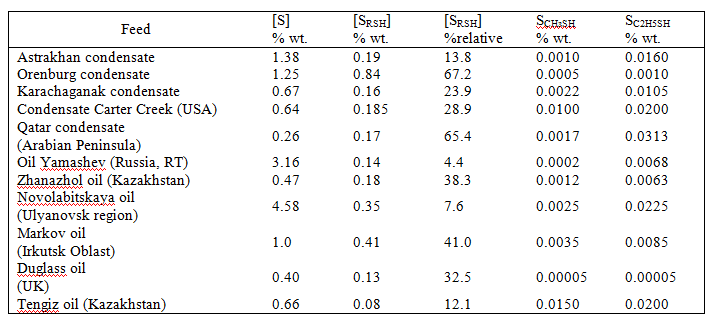
In nowadays processes of oxidative or extractive oil demercaptanization by ACS water-alkaline solutions is solving only the problem of deodorization by the oxidation of light C1-C2 mercaptans in oil to disulfides and to satisfy GOST R51858-2002 requirements and provide acceptable terms of its transportation in Russian Federation or abroad. Thus formed disulfides remain in the oil, i.e. sulfur content in oil is not changed (see Table 2).
Table 2. The composition of sulfur compounds in the Tengiz oil before and after purification by oxidation sweetening.
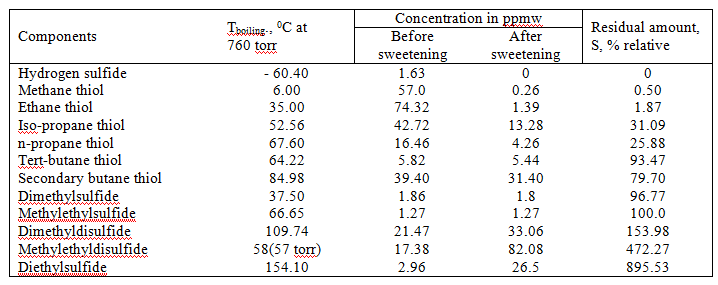
Table 2 shows that the use of oil oxidation sweetening completely oxidized only methyl and ethyl mercaptans. Heavier propane thiol oxidized by 70% and butane thiol – 20%. The residual mercaptans and disulfides formed remain in the oil, causing corrosion and formation of coke deposits at subsequent high-temperature hydrotreatment processing, because disulfides also have low thermal stability and decomposing into the mercaptans and hydrogen sulfide when heated above 200°C.
To facilitate the subsequent processing of high mercaptan gas condensates and light oils we have developed “Demerus” process. The distinguishing feature of Demerus is using of special extractant capable of removing mercap-tans from a high-boiling hydrocarbons. Extractant settling time from the purified gas condensate does not exceed 90 minutes. Aqueous alkaline solution under these conditions is not settled even for 300 minutes (see Table 3).
Table 3. Comparative analysis of efficiency of mercaptans extraction by Demerus extractant and aqueous-alkaline solution.
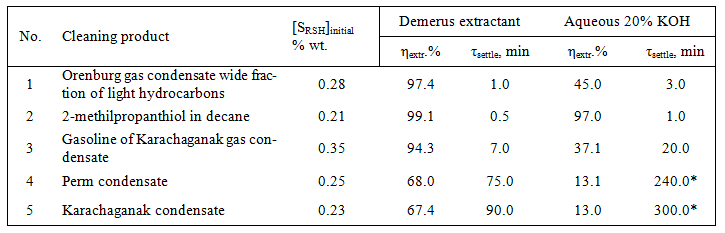
*- aqueous extract in the presence of phenolphthalein is colored.
As seen from Table 3, in case of “Demerus” process gasoline fraction is extracted up to 94% out of mercap-tans (and 37% with the usual alkaline extraction), from gas condensate – up to 67% of mercaptans (against 13%). By extracting mercaptans decreases the total sulfur content in the gas condensate (see Table 4).
Table 4. Treatment effect of mercaptan and total sulfur content at Orenburg gas condensate depend on tech-nologies.
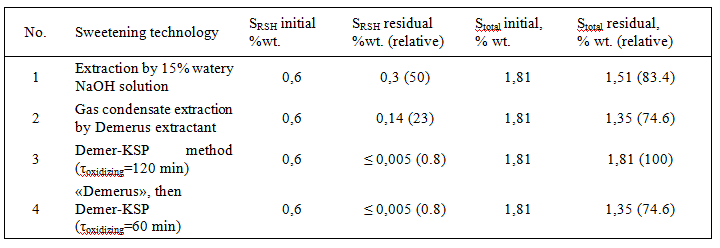
Extractant regeneration is carried out by oxidation of contained mercaptides to disulfides using KSM cata-lyst (Fig. 4).
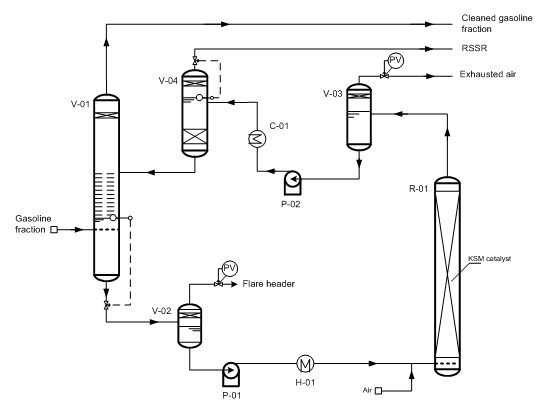
Fig. 4 “Demerus” process for sweetening gasoline fractions.
(V-01) – extractor; (V-02) – degasser; (V-03) – degasser; (V-04) – settler; (R-01) – regenerator; (P-01/02) – pumps; (H-01) – heater; (C-01) – cooler.
At the same time decided important task of obtaining by high mercaptan raw disulfide oil – a valuable petro-chemical product that is an effective agent for sulfuring catalysts of hydrogenation processes and an inhibitor of coke formation during pyrolysis of hydrocarbons.
4. The process of local catalytic oxidation waste decontamination – LOCOS
The essence of LOCOS process is oxidizing by atmospheric oxygen toxic sulfide contained in the wastes to less harmful oxygen-containing compounds – thiosulfate and sodium sulfate or ammonium sulfate and mercaptides – to inert disulfides and further alkiltiosulfonates by the following reactions:
5Na2S + 6O2 + 2Н2О? 2Na2S2O3 + Na2SO4 + 4NaOH (3)
RSNa + О2 ? RSО2Na (4)
Unlike the original sulfides and mercaptides formed thiosulfate, sodium sulfate and alkiltiosulfonates do not have odor and have much higher limit values for plant’s biological wastewater treatment. It is easily achieved at the expense of the actual dilution of oxidized sulfur-alkaline waste with offsite runoff before it enters the biofeedback.
LOCOS process is taking place in the presence of the KSM catalyst at 70?80°C and a pressure of 0.5 MPa. After the complete oxidation of toxic sulfide and mercaptides the waste is sent to the site of neutralization before dis-charging it to the biofeedback (Fig. 6).
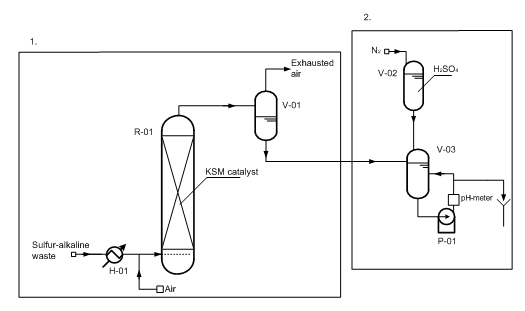
Fig. 6 Sulfur-alkaline waste water decontamination unit, where
1 – hydrogen sulphide oxidation unit,
2 – neutralization unit of oxidized wastes.
(V-01) – degasser; (V-02) – vessel for H2SO4; (V-03) – neutralizing vessel; (R-01) – regenerator; (P-01) – pump; (H-01) – heater.
To reduce water consumption and to provide circulating water supply it is appropriate to provide for desalting oxidized and neutralized wastes by known methods such as reverse osmosis.
In contrast to the process of sulfur-alkaline waste water carbonization or liquid-phase oxidation method, LOCOS process does not require much heat and chemicals and does not pollute the air emissions by hydrogen sul-fide or sulfur dioxide. The alkaline nature of the reaction medium and the relatively low temperature of the process allow using apparatus made of carbon steel. The use of KSM catalyst in LOCOS process can significantly increase the efficiency of sulfur-alkaline waste decontamination in comparison with carbon fiber oxidation catalyst or noncata-lytic method:
1) Reduce the temperature and shorten the oxidation time of sulphides and mercaptides by air oxygen.
2) Provide a deeper oxidation of sulfur compounds and decrease chemical oxygen demand by about 75%.
3) Reduce the amount of toxic mercaptans in the exhaust air.
REFERENCES
FOMIN, V.A., VILDANOV, A.F., MAZGAROV, A.M., LUGOVSKOI, A.I. Implementation of the BBF process sweetening in Ryazan Refinery, Refining and Petrochemicals, No.12, 1987, p.14-15.
Kopilov, A.Yu. Abstract of doctoral dissertation, Kazan, 2010, p.12.
Resolution of the RF Government No.1076 by 30.12.2008.
AKHMADULLINA, A.G., KIJAYEV, B.V., NURGALIYEVA, G.M., SHABAYEVA, A.S., TUGUSHI, S.O., HARITINOV, N.V. Hetero-catalytic demercaptanization of light hydrocarbons. Refining and Petrochemicals, No.2, 1994, p.39-41.
AKHMADULLINA, A.G., AKHMADULLIN, R.M., SMIRNOV, V.A., TITOVA, L.F., EGOROV, S.A. « The expe-rience of heterogeneous catalytic sweetening of MTBE raw in JSC “Slavneft Yaroslavnefteorgsintez », Refining and Petrochemicals, No.3, 2005, p.15-17.
SHARIPOV, A.H. Oxidative desulfurization of mercaptan containing raw materials. Chemistry and Technology of Fuels and Oils, No.4, 1998, p. 9-13.
SHTERBACHENKO, V.I., BAJENKIN, P.M., TOCHILOV, V.A. Refining and Petrochemicals, Moscow, 1979, No. 6, p.23-27.
MAZGAROV, A.M., VILDANOV, A.F. New catalysts and processes for the purification of petroleum and petroleum products from mercaptans, Petrochemistry, 1999, Vol. 39, No. 5, p.371-378.
SHARIPOV, A.H., KIRICHENKO, Yu.E. Kerosene fractions demercaptanization using cobaltpolyphthalocyanine. Chemistry and Technology of Fuels and Oils, No.1, 1998, p. 15-18.
SAMOHVALOV, A.I., SHABALINA, L.N., BULGAKOV, V.A., AKHMADULLINA, A.G., NURGALIYEVA, G.M., SHABAYEVA A.S. Kerosene fraction demercaptanization using the phthalocyanine catalyst, Chemistry and Technology of Fuels and Oils, No.2, 1998, с.43-45.
AKHMADULLINA, A.G., AKHMADULLIN, R.M. New developments and implementations in the field of hydrocar-bon-stock desulfurization.. Chemistry and Technology of Fuels and Oils, 2008, Volume 44, Number 6, Pages 371-378.
BELINSKIY, B.I., BERDNIKOV, V.M. and others. Hydrotreating of mercaptan containing condensate feedstock, Chemistry and Technology of Fuels and Oils, No.3, 2002, p. 8-10.
MAZGAROV, A.M., VILDANOV, A.F., KOPILOV, Yu.P. Resources of mercaptan-containing crude oils and gas condensates, and peculiarities of their processing, Journal of the All-Russian Society of Chemistry by D.I. Mende-leev, р. 67-72, 2004.
PAVLOV, D.V., VARACSIN, S.O., KOLESNIKOV, V.A. Industry circulating water supply. Sanitary engineering, No.2, 2010, p.30-35.
AKHMADULLINA, A.G., KIJAYEV, B.V., KHRUSHCHEV, I.K., ABRAMOV, N.M., NURGALIEVA, G.M. , BEKBULATOVA, A.T., SHABAEVA, A.S. Industrial experience of using heterogeneous catalysts in the oxidative processes of sulfur-alkaline waste water and aqueous process condensates decontamination. Refining and Pe-trochemicals, No.2, 1993, p.19.
AKHMADULLINA, A.G., ORLOVA, L.N., KHRUSHCHEVA, I.K. and others. Mercaptides transformation during the catalytic oxidation with molecular oxygen in aqueous alkaline solutions. Journal of Applied Chemistry, No.1, 1989, p. 53-57.
MURZAKYN, A.R., BADYKOVA, A.D. , KUDASHEVA, F.H. and others. Petrochemical plants industrial methods of sulfur-alkaline waste decontamination. Refining and Petrochemicals. No.1, 2007, p.38-40.
VILDANOV, A.F., LUGOVSKOY, A.I., ARHIREEVA I.A., VASHCHENKO, P.M., LOGINOV, C.A., AYUPOVA, N.R., MAZGAROV, A.M. The new catalyst for the process of sulfur-alkaline waste water decontamination // Che-mistry and Technology of Fuels and Oils. 1990, No.10, p.32.
AKHMADULLIN, R.M., AKHMADULLINA, A.G., AGHAJANIAN, S.I., VASILYEV, G.G., GAVRILOV, N.V. Butane demercaptanization at JSC “LUKOIL Nizhny Novgorod”, Refining and Petrochemicals, 2012.